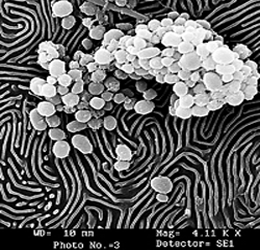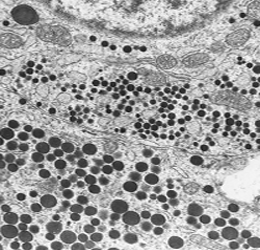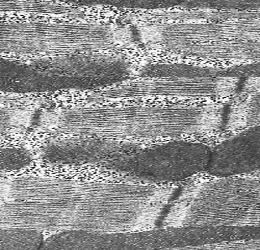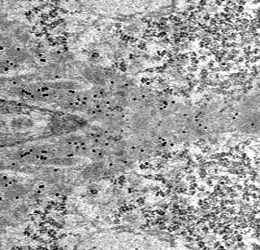
Sputter Coating

· Specimens are placed on a cooled base plate which functions as an anode.
· The cathode, which is the metal (e.g., gold) to be evaporated, is positioned above the specimen.
· The chamber, in which the cathode and anode are located, is evacuated.
· Inert gas such as argon is flooded into the chamber and is again evacuated.
· A negative high voltage is applied to the cathode. This results in glow discharge and argon gas molecules gets ionized.
· The positively charged ion move towards the cathode and strikes on it, thereby releasing Au atoms.
· The dislodged metal atoms due to numerous collisions with gas molecules get deflected several times and hit the specimen at different angles. Thus a uniform thin layer is deposited on the specimen.